DYNAMICS OF MEMBRANE PROTEINS
 KV2.1 VOLTAGE GATED POTASSIUM CHANNELS
KV2.1 VOLTAGE GATED POTASSIUM CHANNELS
The voltage gated potassium channel Kv2.1 plays an important physiological role in numerous cell types and tissues. In particular, in the mammalian neurons, these channels have a protective function in response to stroke suppressing seizures which aggravate brain damage. This critical role is achieved by forming and regulating Kv2.1 surface clusters. We aim at revealing the mechanism of Kv2.1 channel cluster formation using single particle tracking with nanometer resolution in live mammalian cells. A greater understanding of how this channel is dynamically localized may lead to improved treatments for stroke, the third leading cause of death in the United States and a major cause of long term disability. By unraveling the mechanisms underlying surface protein localization and channel surface formation we will improve our knowledge of these molecular processes and advance human health.
We tackle the problem of cell surface domain maintenance using simple growth models and a Fokker-Planck formalism. Surprisingly, our results show that Kv2.1 domains are maintained by fluctuations in endocytosis/exocytosis without any type of feedback or additional regulation of domain size.
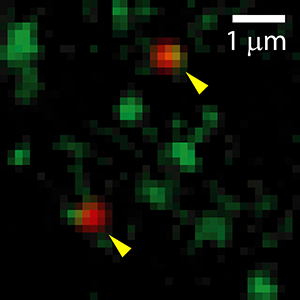
Using multicolor single-particle detection and tracking, we observe that Kv2.1 rapidly recycles between membrane and intracellular compartments and that intracellular Kv2.1-containing trafficking vesicles dock directly at Kv2.1 cell surface clusters. Surprisingly, Kv2.1 channels are not the only molecules that are endocytosed and exocytosed at these clusters, showing that the Kv2.1 clusters are specialized trafficking platforms. This study was the first to define stable cell surface sites for K+ channel delivery in mammalian cells.
We investigate the role of cortical actin in Kv2.1 channel dynamics by using actin inhibitors. Interestingly we see that upon application of actin inhibitors the Kv2.1 channel trajectories regain ergodicity, however still undergo anomalous diffusion. These results show that the actin cytoskeleton plays a dominant role in controlling the CTRW process.
- A.V. Weigel, P.D. Fox, E.J. Akin, K.H. Ecklund, M.M. Tamkun, and D. Krapf, “Size of cell-surface Kv2.1 domains is governed by growth fluctuations”, Biophys. J. 103, 1727 (2012)
- E. Deutsch, A.V. Weigel, E.J. Akin, P. Fox, G. Hansen, C.J. Haberkorn, R. Loftus, D. Krapf, and M.M. Tamkun, “Kv2.1 cell surface clusters are insertion platforms for ion channel delivery to the plasma membrane”, Mol. Biol. Cell 23, 2917 (2012)
- P.D. Fox, C.J. Haberkorn, A.V. Weigel, J.L. Higgins, E.J. Akin, M.J. Kennedy, D. Krapf, and M.M. Tamkun, “Plasma membrane domains enriched in cortical endoplasmic reticulum function as membrane protein trafficking hubs”, Mol. Biol. Cell 24, 2703 (2013)
- A.V. Weigel, M.M. Tamkun, and D. Krapf, “Quantifying the dynamic interactions between a clathrin-coated pit and cargo molecules” Proc. Natl. Acad. Sci. USA 110, E4591 (2013)
- P.D. Fox, C.J. Haberkorn, E.J. Akin, P.J. Seel, D. Krapf, and M.M. Tamkun, “Induction of stable endoplasmic reticulum/plasma membrane junctions by Kv2.1 potassium channels”, J. Cell Sci. 128, 2096 (2015)
- E.J. Akin, L. Solé, B. Johnson, M. el Beheiry, J.-B. Masson, D. Krapf, and M.M. Tamkun, “Single-molecule imaging of Nav1.6 on the surface of hippocampal neurons reveals somatic nanoclusters”, Biophys. J. 111, 1235 (2016)
- G. Sikora, A. Wyłomańska, J. Gajda, L. Solé, E.J. Akin, M.M. Tamkun, and D. Krapf, “Elucidating distinct ion channel populations on the surface of hippocampal neurons via single-particle tracking recurrence analysis”, Phys. Rev. E 96, 062404 (2017)
- M.J. Metz, R.L. Pennock, D. Krapf, and S.T Hentges, “Temporal dependence of shifts in mu opioid receptor mobility at the cell surface after agonist binding observed by single-particle tracking”, Sci. Rep. 9, 7297 (2019)

Krapf Lab © 2018