![]()
Determination of Qin from the Fuel Heating Value
![]()
The higher heating value(HHV) of the fuel will be assumed. This means that all the water products will be liquid. The HHV for each fuel is summarized in Table II, along with the air/fuel ratio for stoichiometric combustion.
Stoichiometric combustion occurs when all the oxygen atoms in the oxidizer react chemically to appear in the products. The oxidizer in this case is the air mixing with the fuel in the piston cylinder. The chemical reaction is given by:
![]()
![]()
Where:
The stoichiometric fuel to air ratio can be calculated with:
FARs = 1 mole of fuel / a1(O2 + 3.76N2)
Table II. Fuel Properties
| Fuel | Chemical Formula | HHV
[kJ/kg] [kg air/kg fuel] |
Air/Fuel Ratio Stoichiometric | Fuel/Air Stoichiometric |
| Methane | CH4 | 55,500 | 17.2 | 0.05814 |
| Propane | C3H8 | 50,300 | 15.6 | 0.06410 |
| Octane | C8H18 | 47,900 | 15.1 | 0.06623 |
| Methanol | CH3OH | 22,700 | 6.5 | 0.15385 |
| Ethanol | C2H5OH | 29,700 | 8.99 | 0.11123 |
| Hydrogen | H2 | 141,600 | 27.2 | 0.03677 |
| Gasoline | C8H15 | 47,300 | 14.6 | 0.06849 |
| Diesel | C12.5H22.2 | 44,800 | 14.5 | 0.06897 |
In order to determine the heat input, Qin, in [J], the mass of the fuel entering the piston has to be calculated.
The equivalence ratio can be expressed as:
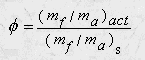
If the equivalence ratio is given the above relation can be solved for the actual mass of the fuel.
![]()
The mass of the air, ma,actual, can be estimated from the displacement volume and the density.
![]()
Where:
The enrgy input, Qin, can be defined as:
Qin = HHVfuel * mf,actual
Two separate fuels can be tested to observe the changes in engine performance with the Engine Performance Applet with Fuel Type. This applet also includes the effect of heat transfer using Woschni's correlation.
![]()
 |
 |